
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License. Image courtesy of Morgan Sproul, Dalhousie College of Pharmacy.


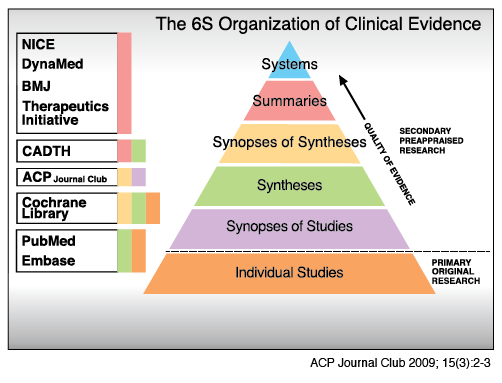
Systems are very detailed, often patient-specific, and link the patient’s conditions to current best practices.
They currently are not very common outside of specialty clinics and/or highly automated departments.

Summaries integrate the best available evidence to provide a full range of evidence addressing all management options for a given health problem, not just one aspect of the problem (as found in single-study resources).
Sources to search include:

Very brief description of original reviews, often with recommendations for practice/decision-making. Sources to search include:

Syntheses include systematic reviews, meta-analyses, scoping reviews, and more. Sources to search include:
For more information, check out our subject guide on systematic reviews.

Provide very brief descriptions of original articles. These are often read as if they are a literature review for the topic, and can also be referred to as critically appraised topics (or CATs). Sources include TRIP, PubMed, and ACP Journal Club. You can also search journals such as Evidence-Based Medicine, Evidence-Based Mental Health, and more.
For more information on CATs, see the subject guide from the University of Alberta on critically appraised topics.

Sources for Individual Studies:
Clinical Trial Registries:
Journal Articles about Clinical Trials: